Trust, but Don’t Verify? FDA’s New Inspection Policy Causing Dramatic Increase in Food Recalls

A century ago, the U.S. federal government made a straightforward decision: If you sell food to Americans, the government will verify that it is safe. Not by reviewing self-submissions. Not by relying on self-representations, but by showing up and seeing for itself. The system relied on Food and Drug Administration officials making surprise inspections and verifying compliance firsthand.
Live Your Best Retirement
Fun • Funds • Fitness • Freedom
This standard held because it worked. It aligned incentives, deterred corner-cutting, and caught problems before they spread. In recent years, though, the FDA has moved away from this standard, and the results have been immediate and remarkable.
Prior to 2021, food recalls were rare. Then the FDA quietly implemented “announced” and “remote inspection” draft policies in January 2021, right at the beginning of the Biden presidency, to align with its full-time/maximal “work at home” policies during the COVID-19 pandemic. This caused FDA recalls to surge.
What began as a temporary government program ossified into a permanent FDA policy in June 2025, despite the risk from COVID-19 having long been gone and FDA employees returning to the office. However, facilities that once expected surprise FDA inspections began operating under a regime defined by manufacturer-supplied documentation, self-submitted testing, and remote, scheduled (rather than unannounced) FDA interactions.
The result? Food recalls are no longer occasional disruptions. They are now a regular feature of our FDA. Recall data shows a clear, sustained rise, with products contaminated by pathogens, foreign materials, or basic cleanliness and sanitary failures reaching consumers, and only then being recalled.
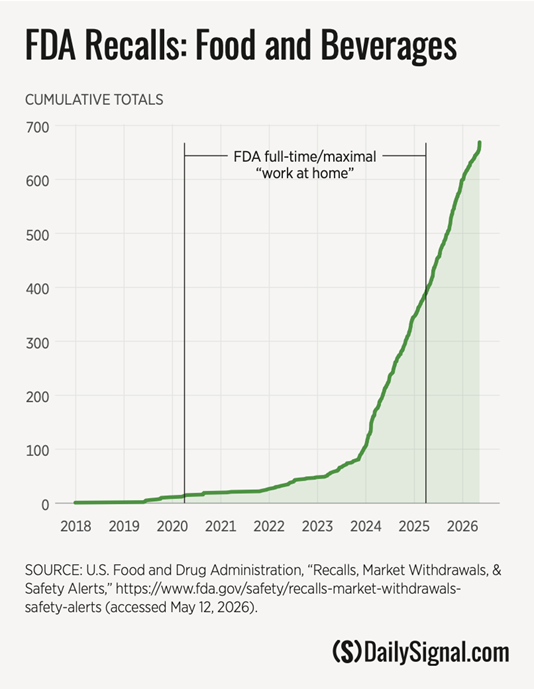
The direction of the graph is clear. The slope is steep and reflects a policy change implemented by FDA leadership that favors trusting manufacturers and detecting problems after products are delivered to store shelves and consumers.
And these “official” FDA recalls are just the tip of the iceberg. Only a small fraction of food-related illnesses is reported, per independent studies, plus the Centers for Disease Control and Prevention.
Teresa Murray, a consumer watchdog at U.S. Public Interest Research Group states: “There’s a decent chance that you or someone close to you has become ill from food poisoning during the last few years yet didn’t realize it unless the sickness was bad enough to see a doctor.”
The cause is obvious and demonstrable.
In any regulated system, behavior follows incentives. When the likelihood of unannounced, physical inspection evaporates, noncompliance follows. Over time, the change expressed itself in outcomes.
Problems that would have been identified at the point of production are now only discovered well after distribution—after Americans may have already consumed recalled foods—when the only remaining action is a post-marketing recall.
The data tracks that shift with uncomfortable clarity. Food recalls alone have skyrocketed, with dozens of events in single months. Per the FDA’s published data, contamination of recalled products has included findings of botulinum, Salmonella, Listeria, E. coli, and other foodborne bacteria. These incidents reflect a fundamental lack of basic food-handling and cleanliness protocols, and they have the potential to be responsible for sickening or killing Americans.

Today’s food recalls are not outliers, they are almost daily occurrences. Hardly a weekday goes by that the FDA doesn’t issue a recall for something, according to a search of its main FDA.gov recalls webpage and its X account (formerly Twitter). The account lists over 7,400 postings, per the upper left-hand corner of the image above (denoted with a green arrow).
The current administration inherited this structure along with a backlog of inspections that built up during the pandemic. However, it also inherited the responsibility to keep our food supply safe. Thus far, it has not done so. It’s bad enough that even the U.S. Government Accountability Office publicly scolded the FDA as recently as February 2026 about not effectively overseeing America’s food safety.
It’s more than a little ironic, because the now-exited FDA Commissioner Marty Makary promised that the safety of our foods would be emphasized. He had frequently and repeatedly stated that “people forget the ‘F’ in FDA stands for food,” and “we want to focus on food,” and “It’s time to tell people the truth about food.”
Unfortunately, those promises never translated into FDA regulatory policy action when it came to postmarketing safety recalls.
Remote testing methodologies remain in place, the inspection cadence has not been reestablished, and within the agency, continuity has been uneven, with senior roles turning over multiple times and experienced personnel exiting at a concerning pace.
Food safety does not tolerate policy drift. It requires a stable, predictable enforcement posture and a clear signal to the market that standards will be verified, and consumers protected.
The solution is clear:
Reestablish unannounced, in-person inspections as the default across high-risk food facilities, both domestic and foreign. Treat manufacturer-submitted documentation as supporting material, not the basis of compliance. Clear the inspection backlog on a defined schedule and publish regular progress reports. Concentrate resources where the risk is highest and maintain a visible presence that resets expectations across the supply chain.
Measure performance where it matters. The relevant metric is not how many recalls are issued. It is how many are avoided because of competent, independent inspection and testing.
None of this requires new authority or novel theory. The statutory framework already exists. The expertise exists. The operational playbook is well understood. It is a question of discipline and execution by leadership.
An America-first MAHA approach to food-safety policy begins with a simple commitment: what is sold in this country must meet standards that are independently verified, not unscientifically assumed.
That commitment is enforceable. It has been enforced before. It must be enforced again.
When it comes to America’s food safety, the FDA does not need to reinvent itself. It just needs to show up to work again.
What's Your Reaction?
 Like
0
Like
0
 Dislike
0
Dislike
0
 Love
0
Love
0
 Funny
0
Funny
0
 Wow
0
Wow
0
 Sad
0
Sad
0
 Angry
0
Angry
0

























Comments (0)